|
5/7/2023 0 Comments Spike proteins 
As already noted, the artificial microstructure is produced under significantly off-equilibrium conditions, compared to the near-equilibrium conditions that prevail during assembly of the native protein coat. In tensile tests conducted on dry fibre, the breaking strength is ~ 0.3 GPa, the stiffness ~ 4 GPa, and the elongation to failure ~ 30 % ( Hudson 1997). The uniaxial mechanical properties of fibre spun from genetically engineered analog of spike protein have been disappointing. However, there is no evidence that fibres spun from this material contain trimeric box beams in their hierarchical microstructure ( Gillespie et al. Genetically engineered model polymers based on multiple consecutive copies of the principal repeated sequence in native adenovirus spike protein can self-assemble into fibrillar structures, which in turn can form a liquid crystalline phase in solution ( O’Brien 1993). Hydrophobic bonding is presumed to be important in stabilising their structure, since 70% of the amino acid residues are hydrophobic in representative cases ( Estes and Cohen 1989). There may also be significant intrinsic differences between the architecture of spikes from different types of virus. However, these non-natural fibres are assembled under significantly off-equilibrium conditions, and so cannot be assumed to have the same internal structure as the native material. Attempts have been made to produce simple genetically engineered analogs, and to spin fibres for analysis by microscopy, diffraction or spectroscopy. Typically, dimensions do not exceed a length of 30 nm or a width of 5 nm. Structural characterisation is hampered by the small size of individual spikes. Compressive stiffness in this case is ascribed to opposite twists at the coil and supercoil levels. 1987) describe the spike as having a triad of helices at its core – somewhat like the collagen triple helix, but with a continuous space enclosed between the three molecular helices. Three cross-ß-sheets are thought to interact to form a hollow trimeric box beam ( Figure 8.10) that resists buckling in compression. 1997) and human adenovirus ( Chatellard and Chroboczek 1989 Hoess et al. It is not universally accepted that the structure is based on cross-ß-sheets, although that model is well supported for both rotavirus ( Isa et al. 1987 Delmas and Laude 1990 Stouten et al. 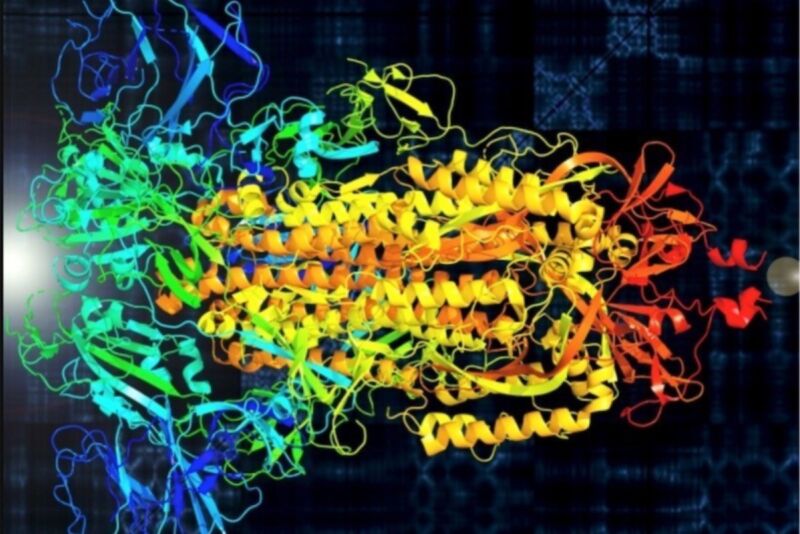
8.2.4.2 Higher-order structure.Ĭontroversy still surrounds the detailed higher order structure of viral spikes, and there appears to be considerable diversity of structure between virus types ( Green et al. 1995 Sikkema 1998 ), the hierarchical structure of spike proteins deserves careful attention. Because compressive strength and stiffness have been a long-term elusive goal of polymer science ( Santhosh et al. Hollow spikes on the capsid fulfil the latter two roles, from which it has been deduced that they must have unusually high strength and stiffness in axial compression ( O’Brien 1993). The structural characteristics of virus coats are highly relevant to virus propagation, and therefore are the subject of intensive study – the coat must protect the nucleic acid (genetic) contents, be resilient against impact, be capable of broaching the outer wall of a target cell, and provide a secure pathway for conducting nucleic acid into the target. An analogy is provided by the “do loops” used in computer programming. The relatively small amount of genetic information stored in a virus means that simple and / or repeated motifs occur in the protein that makes up the surrounding capsid (coat). Emily Renuart, Christopher Viney, in Pergamon Materials Series, 2000 8.2.4 Viral spike protein
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
